Purpose:
- The purpose of the flame test lab is to observe the characteristics colors produced by certain metallic Ions when vaporized in a flame. Identify unknown metallic Ions by means of its flame test.
Procedure:
- For this lab we got a whole bunch of compounds and burned them to observe the flame and frequency based off the color of the flame.
Pre-Lab Questions:
- What color of light is the lowest in energy?
- Red
- What color of light is the highest in energy?
- Violet
- What color of light has the highest frequency?
- Violet
- What color of light has the lowest frequency?
- Red
- Explain "Ground State"
- Ground State is when there is an electron(s) with the lowest possible energy levels.
- How are electrons "excited"
- They are given energy
- What does it mean when the electrons are "excited"?
- They are receiving all the energy from the heat or whatever is feeding it the energy.
- In your own words, write a short explanation on how an electron absorbs energy and re-emits it as light and why different elements have different spectra.
- The electron absorbs the energy in this case it would be the heat and it re-emits the energy when the element is dying.
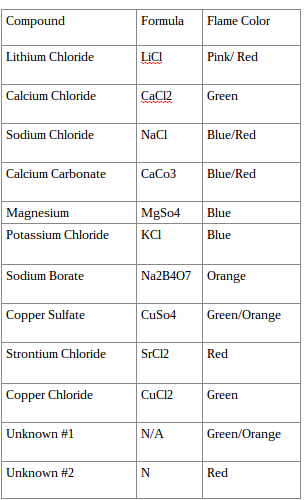
Data:
Discussion Of Theory:
Error Analysis:
Post-Lab Questions:
Conclusion:
- A theory that I have for this lab is that if you were to add unnecessary drops to the compound and you just add a lot more then needed if it has an affect to the color of the flame and then the lab wouldn't be accurate. This theory is relevant because it is gonna help you get an accurate lab and help you find a very accurate conclusion to this lab. This is also a theory because maybe if you added just the right amount of drops would the flame would be a pure color instead of like a blue and red maybe it would just be red or some color close to that.
Error Analysis:
- There were no errors that my partner and I faced with during this lab.
Post-Lab Questions:
- Why is it important to test the flame color of menthol without any compounds dissolved in it?
- It is important t know the color of it because if the compound is burning and it has a mix of colors you have to take into consideration that it could just be the Menthol burning and not the actual compound.
- List the colors observed in this lab from highest to lowest in energy.
- Blue,Green, Orange, Red
- List the colors observed in this lab from least to greatest from highest frequency to lowest frequency.
- Blue, Green, Orange, Red
- List the colors observed in this lab from shortest wavelength to longest wavelength.
- Red, Orange, Green, Blue
- What is the relationship between energy, frequency, and wavelength.
- The relationship is that they all have to do with something that regards to the color of the flame.
- Based on the results of your experiments what metal was found in your unknowns?
- I would assume that it would be copper because of the flame color and the way it looked after the flame had finally died out.
- Do you think we can use the flame test to determine the identity of unknowns in a mixture? Why or why not?
- I believe it will help us make an educated guess on what it might be, I do not think the flame test can help us find exactly what it is all the time though.
- Why do different chemicals emit different colors of light?
- Because all the chemicals have different amounts of energy.
- Why do you think the chemicals have to be heated in the flame first before the colored light is emitted?
- Because the chemical needs to absorb the energy to emit the color of light.
- Most salts contain a metal and non-metal. Look at the compounds we tested and explain how we can be sure that it is the metal atoms that are responsible for the colors you see.
- You can tell because if you look at the data most of the metals were colored but were not red or blue like the non-metals, so I think you will be able to tell based off the color of the flame.
- Colorful light emissions are applicable to everyday life. Where else have you observed these light emissions? Are these light emissions applications related? Explain
- I have observed these light emission at bonfires where at some points when the fire would get bigger it would sometimes change colors.
- Can you think of a way in which to use the flame test? Please describe below
- I'm not really sure. Can you tell me if there is a way we can use the flame test?
Conclusion:
- In conclusion, the flame test lab was meant to teach us about the flame colors as well as what the flame colors mean as well as the frequency levels based off the color of the flame.