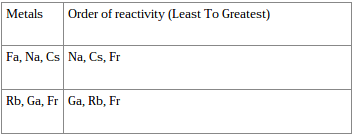Purpose:
Pre-Lab Questions:
Part 1: Trends in Properties within Groups
Procedure:
- To explore the reactivity trends of metals in groups and periods of the periodic table.
Pre-Lab Questions:
- What are groups? What are periods? What are families?
- The groups of the periodic table are the vertical columns. The periods of the periodic table are the horizontal rows. The groups of the periodic table all share similar properties and traits.
- In general, where are metals located on the periodic table?
- The metals are on the left of the metalloid boundary.
- What are the names of the two metal families?
- The 2 metal families are the alkali metals as well as the alkaline earth metals.
- What element is in Group 3 and in the 3rd Period?
- The element Gallium is in the 3rd group and the 3rd period of the periodic table.
Part 1: Trends in Properties within Groups
Procedure:
- Acquire test tubes.
- Place small strip of magnesium in a test tube.
- Label the test tubes.
- Fill beaker with paper get disposable pipet.
- Get sample of calcium from Charlene.
- Use Pipet to cover sample.
- Observe reaction carefully, then enter data.
- Dump sample into hand over sink, drop test tube into beaker, wash out test tube.
Part 2: Activity Series of some metals in HCI
Procedure:
Procedure:
- Acquire 1 more clean test tube.
- Label the test tubes.
- One person goes to acquire the HCI.
- Acquire the Zinc and Tin from Charlene.
- Cover sample with dilute HCI drop by drop.
- Observe the reactions, fill in data table.
- Bring samples to fume hood, then poor into waste bottle.
Analysis Questions:
- What can you conduct about the reactivity of metals as you move down the column or group in the periodic table.
- As you move down the columns in the periodic table, the reactivity slowly decreases.
- What can you conclude about the reactivity of metals as you move across a period?
- I noticed that when I move across the period the reactivity decreases.
- Arrange each set of the following metals in order from least to most reactive using conclusions you just made.
- Refer to Chart Below for answer.
- What is the most reactive metal in the periodic table? Explain. Would you expect this metal to be found in its unreacted elemental form? Explain.
- I would expect Francium to be in its unreacted elemental form because it would probably be dissolved if it were to react with something, because Francium has the greatest atomic radius and is extremely reactive.
- The following is a reactivity chart that was found on the internet.
- Refer to Picture to see chart.
Discussion of Theory:
Post Lab Questions:
Conclusion:
- We found a couple trends in this lab, we found that as you move down the columns in the periodic table the reactivity decreases slowly, and we also found another trend and it was that wen you move to across the periodic table the activity also decreases.
Post Lab Questions:
- What might be a reason for the difference in behavior between magnesium and calcium when placed in water? (Hint: lithium, sodium, and potassium are also progressively more reactive).
- I think that it had a different reaction to calcium because calcium was a powder and water needs a lot of time to break a solid down and magnesium is a solid.
- List the four metals from most reactive to least reactive. Use data from lab to support answer.
- I would say that Zinc had the biggest reaction because most of the stuff dissolved but none of it smoked up like Zinc did. Next would be Magnesium because it dissolved and didn't smoke up. Next calcium because it turned into a liquid that didn't stick to the walls of the test tube. Then tin because it was least reactive and didn't even have a reaction.
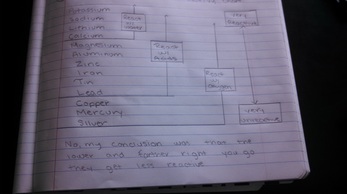
- In general, is there a relationship between the locations of metals on the periodic table and their relative activity? Explain why.
- Yes, they are organized by groups that are made based on the properties that they have. The elements that have a lot in common get put Ito the same groups.
- When elements are organized in the periodic table, various trends appear, describe some of the trends that you learned about from this lab.
- Elements that are easily dissolved in HCI, some got broken down by the HCI and some just don't react to the HCI at all.
Conclusion:
- In conclusion, we learned about what reactions magnesium and calcium would have in water, we also learned about what Tin, Zinc, and Magnesium and how they react to HCI, then we collected all of our data on charts that you can see if you scroll up.